
 Scroll to Explore
Scroll to Explore
Highlight Optipharm records by the numbers
Xenotransplantation
A leader in supporting human welfare
through the recreation of the value of life
Xenotransplantation is a process undertaken to resolve the problem of organ shortage, in which organs, tissues,
or cells from the animal of one species are transplanted to an animal of another species.
VLP
Vaccine
Preparation of a safe vaccine antigen
without the capacity for infectivity and proliferation
With insect cell based VLP vaccine antigen production technology (OptiVLP™), we established a system in which it takes less than six months to complete a process spanning from selecting target genes to securing seeds for vaccine production.
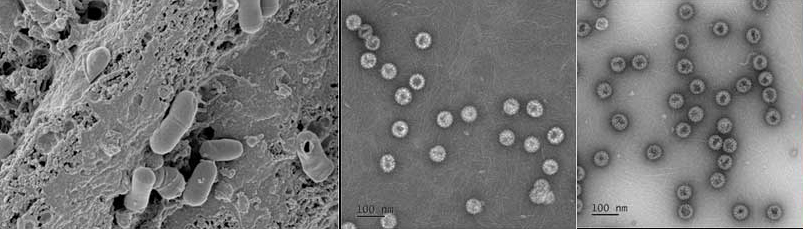
Bacteriophage
Safely and effectively
removes pathogenic bacteria
We secured about 400 excellent bacteriophages that are effective against pathogenic bacteria,
applied for a patent, and carried out a number of studies, which ultimately led to a successful
commercialization of bacteriophage products.
Animal
Diagnosis
‘Useful life resource’
obtained through
animal diagnosis
process
By approaching the issue from a range of
perspectives, we are making a more
professional and accurate diagnosis of
animal diseases.
Medipig We provide animal models for biological and medical research for the treatment of incurable human diseases
Through the research and development of miniature pigs,
we are mainly involved in the sale of miniature pigs for
laboratory animals and contract research organization (CRO)
to contribute to the improvement of human health and welfare.
Notice
-
11 14CERTIFICATION _ OCCUPATIONAL HEALTH AND SAFETY MANAGEMENT SYSTEM (ISO 45001)
-
06 30[Update] The paper on the development of transgenic animals _ Scientific Reports
Hello, OptiPharm's homepage.administrator. The paper on the development of transgenic animals has been finally approved for publication in Scientific Reports, a sister magazine of the Nature journal, and we are informing you as follows.- A desirable transgenic strategy using GGTA1 endogenous promoter-medicated knock‑in for xenotransplantation model- Related Links: https://www.nature.com/articles/s41598-022-13536-z.pdf
-
03 16Opening a new website
Hello, We are currently preparing to open a new website. We will do our best to open as soon as possible. Thank you.



